Mineral froth flotation
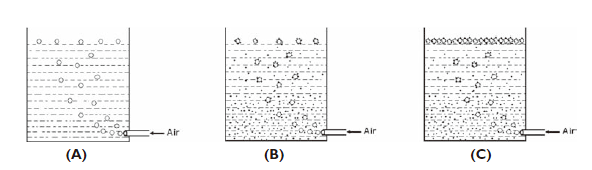
Sinonine deeply know that in a beaker of
water, if air is introduced from the bottom, air bubbles are produced and rise to the
surface of the water as the density of an air bubble is much less than that of water
(Figure.1 A). Similarly air bubbles also rise if air is introduced in a pulp containing
mineral particles. If a mineral particle of high density adheres to the air bubble, the
air bubble along with the mineral particle rises to the surface because the apparent
density of the air bubble and the adhered mineral particle is less than that of water. If
many mineral particles are adhered to the air bubble, still the air bubble rises to the
surface as the apparent density of the air bubble and adhered mineral particles is less
than that of water due to the relatively large volume of the air bubble Figure 1 B). This
concept is the basis for froth flotation operation.
To use this
concept or phenomenon, the following are required:
1 Method to make
the mineral particles adhere to the air bubble.
2 Method to keep
the air bubble alive when it reaches the surface of water. It is known that the air
bubble collapses when it reaches the surface of water. Whenit is collapsed,
the adhering mineral particles are dropped into the water. So the air bubble must be
kept alive on the surface of the water for a sufficient length of time for it to be
removed from the surface.
The following are
the methods adopted in practice:
1 To make the
mineral particles adhere to the air bubble whilst it is rising through the pulp, the mineral
particles are treated with suitable chemicals to acquire adhering properties.
2 To prevent the
air bubble from collapse, some other chemicals are used to prevent the collapsing of
the air bubble or in other words to increase the life of the air
bubble.
The required
mineral particles after adhering to the air bubble float to the surface along with the air
bubble. The aggregation of several such mineral adhered air-bubbles forms the froth on
the surface of the pulp (Figure1 C). Hence this operation of mineral separation
is named Froth flotation.
Flotation is a
method of separating the minerals in a relatively finely divided state.It utilises the
differences in physico-chemical surface properties of particles of various minerals.
Figure 1 Process of rising air bubbles and forming
froth.
This
method can only be applied to relatively fine particles (less than 150 ìm).If the particles
are too large, the adhesion between the particle and the bubble is less than the weight of
the particle and the bubble drops the mineral particles. The air bubbles can only stick to
the mineral particles if they can displace water from the mineral surface. This can
only happen if the mineral is water repellent or hydrophobic.
Air bubbles, after
reaching the surface, can continue to hold the mineral particles if they can form a stable
froth. If not, air bubbles will burst and drop the mineral particles. In order to achieve
the favourable conditions for froth flotation, the pulp is treated with various chemical reagents known as flotation reagents. The chemicals used for treating the mineral
particles to make them to adhere to the air bubbles are called collectors and the chemicals
used to increase the life of the air bubbles are called frothers.
The majority of
the ores mined at present require fine grinding for a high degree of liberation of
valuable minerals, and thus the flotation becomes the only possible means of
beneficiation for higher grades and recoveries. Flotation treatment is extensively
applied to the concentration of metalliferous minerals, both sulphides and oxides. About
90% of the world’s important ores of lead, zinc and copper are upgraded by
flotation operation. The flotation technique is also extensively used in upgrading of
non-metallic ores like graphite, limestone, fluorite, fluorspar, clay, rock phosphate and
coal.
FLOTATION
REAGENTS
Flotation reagents
are substances added to the ore pulp prior to or during flotation in order to make
it possible to float valuable mineral particles and not to float the gangue mineral
particles. Important flotation reagents are collectors, frothers, depressants, activators and pH
regulators.
A Collector is a chemical reagent, either an acid, base or
salt, and is hetero-polar in nature; the polar
part of it has an affinity towards a specific mineral and the non-polar part
has an affinity towards an air bubble. A small amount of collector is added to the pulp and
agitated long enough that the polar part is adsorbed on to the mineral to be floated
whilst the non-polar part is oriented outwards and makes the surface of mineral particles
hydrophobic. The collector increases the contact angle of the valuable mineral particles.
Collectors are
broadly classified as anionic, cationic and oily collectors. Anionic and cationic
collectors are ionizable organic compounds. They are said to be an anionic or cationic
collector whether the ion that carries hydrocarbon group is anion or cation. Oily
collectors are oily liquids which spread out thinly on solid surfaces to cause bubble
attachment. Xanthates,
Dithiophosphates (known as Aerofloats), Dithiocarbamates, Fatty acids and soaps are the
important anionic collectors. Xanthates are the most widely used collectors for
flotation of sulphide minerals. The general formula of sodium or potasium xanthate
is:
The R group, in
the case of ethyl and isopropyl xanthates, are C2H5 and C3H7 respectively
and is shown
below:
The general
formulae for other anionic collectors are:
Dithiophosphates
and dithiocarbamates are used in the flotation of sulphide minerals; carboxylic collectors are used for flotation of non-sulphides and nonsilicates. Cationic
collectors are used for oxide and silicate minerals including quartz.
Amines are the
most commonly used cationic collectors. Oily collectors normally used are petroleum
products, blast furnace oils, coal-tar and wood-tar creosotes. They are used in flotation
of oxidized metalliferous ores and gold ores.
A Frother is a chemical reagent and is heteropolar in nature;
the polar part of it has an affinity
for water and the non-polar part has an affinity for gas or repulsion for water. The
frother acts upon the gas water interface. The addition of a frother decreases the
surface tension of water and increases the life of bubbles produced. The main objective
of a frother is to permit the production of a sufficiently stable froth to hold the
mineral particles that form a network around the bubbles until they are removed from
the flotation unit.
As a result of the
addition of a frother, the gas bubbles, formed under the surface of the water, are
more or less completely lined with a monomolecular sheath of frother molecules
which allows each bubble to come in contact with other bubbles.
This forms a
froth. Thus a froth is simply a collection of bubbles.Cresylic acid and
pine oil are the most widely used frothers. A wide range of synthetic frothers
are now in use in many plants. Methyl Iso-Butyl Carbinol (MIBC) is most important
among the synthetic frothers. The following are the chemical formulae of these organic
reagents.
Eucalyptus oil,
camphor oil and sagebrush oil are used when they are more cheaply available
than the common frothing agents.
To have an
independent control, the frother should not have a collecting property.
The reagents
having both frothing and collecting properties are known as frothercollectors.
The compounds like
fatty acids, sulphonates and amines which are in use as collectors have
also frothing properties. Kerosene is a frother-collector used in coal flotation.
Other chemical
reagents, depressants, activators and pH regulators, called modifiers, are used
extensively in flotation to modify the action of the collector, either by intensifying or
reducing its water – repellent effect on the mineral surface. Thus they make collector
action more selective towards certain minerals.
Depressants
are inorganic
chemicals. They react chemically with the mineral particle surfaces to
produce insoluble protective coatings of a wettable nature making them non-floatable
even in the presence of a proper collector. Thus formed protective coatings prevent
the formation of collector film. Sodium or potassium cyanide is a powerful depressant
for sphalerite and pyrite. A combination of sodium cyanide and zinc sulphate is
more effective in depressing zinc sulphide minerals, sphalerite and marmatite. They
also have a depressing action on pyrite. Lime is sometimes used to depress pyrite
in sulphide flotation. Sodium or potassium dichromate is used to depress galena.
Sodium silicate is
extensively used for the depression of silicates and quartz. Sodium silicate is
much used as a dispersant
for removing
slimes from particle surfaces of sulphide
minerals. Sulphuric acid is used to depress quartz in soap flotation.
Lactic acid is a
powerful depressant in iron sulphide flotation. Metaphosphates are used as
depressants for non-silicates like barite, fluorite and calcite.
Activators, generally inorganic compounds, can
modify the surface of non-floatable or poorly
floatable mineral particles by adsorption on particle surface so that the collector may film
the particle and induce flotation. An example of this is the use of copper sulphate
in the flotation of sphalerite. Copper sulphate dissociates into copper ions in
solution and copper sulphide is formed at the surface of sphalerite. Then it reacts
with xanthate and forms insoluble copper xanthate which makes the sphalerite surface
hydrophobic. In the flotation of lead-zinc ore, after lead flotation, the sphalerite is
activated with copper sulphate and floated. Copper sulphate also activates
depressed pyrite when added in sufficient quantity.
Table1 Quantities of
flotation reagents.
Sodium sulphide is
used to activate oxide minerals of lead, zinc and copper such as cerussite,
smithsonite and malachite. As sodium sulphide imparts sulphide surface to the mineral
particles to facilitate for collector coating, this activator is also known as sulphidizer. Sodium sulphide is also used to float previously
depressed pyrite.
It has dispersing
and depressing effects on sulphide minerals when added in large quantities.
pH
regulators are
used to modify the alkalinity or acidity of a flotation circuit or in other words to
control the pH of the pulp. The pH of the pulp has an important and sometimes very
critical controlling effect on the action of the flotation reagents.
Common pH
regulators are lime and soda ash for creating alkaline conditions, sulphuric and hydrochloric
acids for creating acidic conditions.
The quantity of
reagents used in flotation varies from ore to ore and day to day or hour to hour
for one ore. Small quantities of reagents are normally required. Low and high
quantities of the reagents are of no use. In certain cases, increasing
quantities results in other
effects. For example, use of increased quantities of collector tends to float other
mineral particles (other than required) also. Table 1 shows the approximate quantities of
various reagents used in flotation.
TYPES
OF FLOTATION
As a mineral is
selected and floated in flotation, it is called selective flotation. As a required mineral
is selected and floated, it is also called direct flotation. When an unwanted mineral
is selected and floated, in which case, sink is the required product, it is called reverse flotation. When an ore contains two or more
valuable minerals, one valuable mineral
is selected and floated first and second valuable mineral is floated from the tailings.
This flotation is called differential flotation. Alternatively, when all the valuable
minerals are selected and floated, it is called bulk flotation. The individual minerals are
selected and floated one after the other from the floats.
FLOTATION
MACHINES
A flotation machine
is the equipment used to carry out flotation operation. It provides the hydrodynamic
and mechanical conditions which effect the separation. Basically the flotation
machine must include:
1 Means for
receiving the pulp.
2 Means for
agitation and mixing the pulp.
3 Means for
settling and discharging the pulp.
4 Means for air
introduction and dispersion.
5 Means for
discharging the froth.
The most commonly
used flotation machines are of two types, namely mechanical type and pneumatic
type. In a mechanical type flotation machine a mechanically driven impeller
agitates the pulp and disperses the incoming air into small bubbles.
The air may be
drawn in by suction created by the impeller or may be introduced to the base of the
impeller by an external blower. These flotation machines are often composed of
several identical cells arranged in series in such a way that one cell receives the
de-frothed pulp (tailing of preceding cell) as feed. Such a series of cells are called a bank. The
Denver sub-aeration machine (Figure 2) is well known cell-tocell machine.
The pulp from the
weir of the preceding cell flows through the feed pipe on to the rotating
impeller. The positive suction created by the impeller draws air through a hollow standpipe,
sheared into fine bubbles by impeller and intimately mixed with the pulp. The diffuser
arranged around the impeller prevents the agitation and swirling of the pulp above the
impeller. The stationary hood above the impeller prevents the pulp in the cell
having direct contact with the impeller. The mixture of bubbles and pulp leaves the
impeller and bubbles ascend with their mineral load to form the froth. As the bubbles move to
the pulp level, they are carried to the overflow lip by the crowding action of
succeeding bubbles and removed by froth paddles. Pulp from the cell flows over the
adjustable weir on to the impeller of the next cell. Particles which are too heavy to flow over
the weir are by-passed through sand relief ports.
Figure 2 Denver sub-aeration cell
In an open-flow
type machine, intermediate partitions and weirs between cells are eliminated. The
pulp is free to flow through the machine without interference. In pneumatic
machines, air is introduced through the porous bottom of the cell.
The air used in
these machines not only creates aeration and produces froth but also
maintains
suspension by circulation.
FLOTATION
OPERATION
The flotation
operation is generally carried out in three stages namely roughing, scavenging
and cleaning,
called a flotation circuit (Figure 3). Each stage consists of a bank of cells
and the number of cells in a bank is primarily depends upon the residence time of
the pulp in the cells and the required throughput. The reagent conditioned feed pulp is
treated in a first bank of cells called roughers. The tailing from the rougher cells,
which may still contain some valuable mineral particles, is treated in another bank of
cells called scavengers. The concentrate from the rougher cells is further treated in
a bank of cells called cleaners to obtain high grade final concentrate.
The scavenger
concentrate and cleaner tailing are re-fed to the rougher bank to
be treated with
the fresh feed pulp. The scavenger tailing is the final tailing. It is to be noted that the
pulp is treated in a conditioner, called conditioning, with necessary reagents like depressant and collector, prior to flotation in order to convert the mineral particles to
respond readily in a flotation cell. Conditioning the pulp reduces its residence time in
the flotation cell and hence the capacity of the cell increases.
Figure 3 A typical flotation circuit
COLUMN
FLOTATION
The column flotation technique, which is a recent development, uses the principle of counter current
wash-water flow for better separation particularly when operating on fine
materials. The flotation column is a simplest form of pneumatic type flotation machine. It
consists of a tall cylindrical column having the height to diameter ratio of more than 10
(Figure 16.5). The reagent conditioned feed pulp enters somewhat at the middle of the
column. Compressed air is admitted through a distributor near the bottom of the column. In
the section below the feed point, called recovery section, the feed pulp travels downwards
against the rising air bubbles. Valuable mineral particles adhere to the air bubbles
and are transported to the top part of the column, called washing section.
Gangue mineral
particles that are loosely attached to the bubbles are washed down by water sprays and
only clean froth rises and discharged through annular trough.
The basic
advantage of column flotation is the production of high grade concentrate without the loss
of recovery. A single flotation column can replace five to six stages of operations
involving conventional cells and yet achieve better performance. There are considerable
savings in reagent requirement. The column occupies less floor space.
Figure
4 Flotation column
on fine materials.
The flotation column is a simplest form of pneumatic type flotation machine. It
consists of a tall cylindrical column having the height to diameter ratio of more than 10
(Figure 4). The reagent conditioned feed pulp enters somewhat at the middle of the
column. Compressed air is admitted through a distributor near the bottom of the column. In
the section below the feed point, called recovery section, the feed pulp travels downwards
against the rising air bubbles. Valuable mineral particles adhere to the air bubbles
and are transported to the top part of the column, called washing section.
Gangue mineral
particles that are loosely attached to the bubbles are washed down by
water sprays and
only clean froth rises and discharged through annular trough.
The basic
advantage of column flotation is the production of high grade concentrate without the loss
of recovery. A single flotation column can replace five to six stages of operations
involving conventional cells and yet achieve better performance. There are considerable
savings in reagent requirement. The column occupies less floor space.
FLOTATION
PRACTICE OF SULPHIDE ORES
Flotation
operation was initially developed to treat the sulphides of lead, zinc and copper. In a
typical flotation practice of lead-zinc ore, the different reagents used are sodium cyanide and
zinc sulphate as depressants for pyrite and sphalerite, potassium ethyl xanthate as
collector for lead circuit to float galena, sodium isopropyl xanthate in zinc circuit to
float sphalerite, copper sulphate to activate already depressed sphalerite in zinc
circuit, lime as pH regulator, and crysilic acid as frother in both circuits. In some
plants, Methyl Iso-Butyl Carbinol (MIBC) is used as a frother. For the flotation of
chalcopyrite (copper mineral in most of the copper concentrators), sodium isopropyl
xanthate and pine oil are the collector and frother respectively in most of the
plants. Soda ash is used as a pH regulator.
sinonine can also provide sand washing plant epc.










评论
发表评论